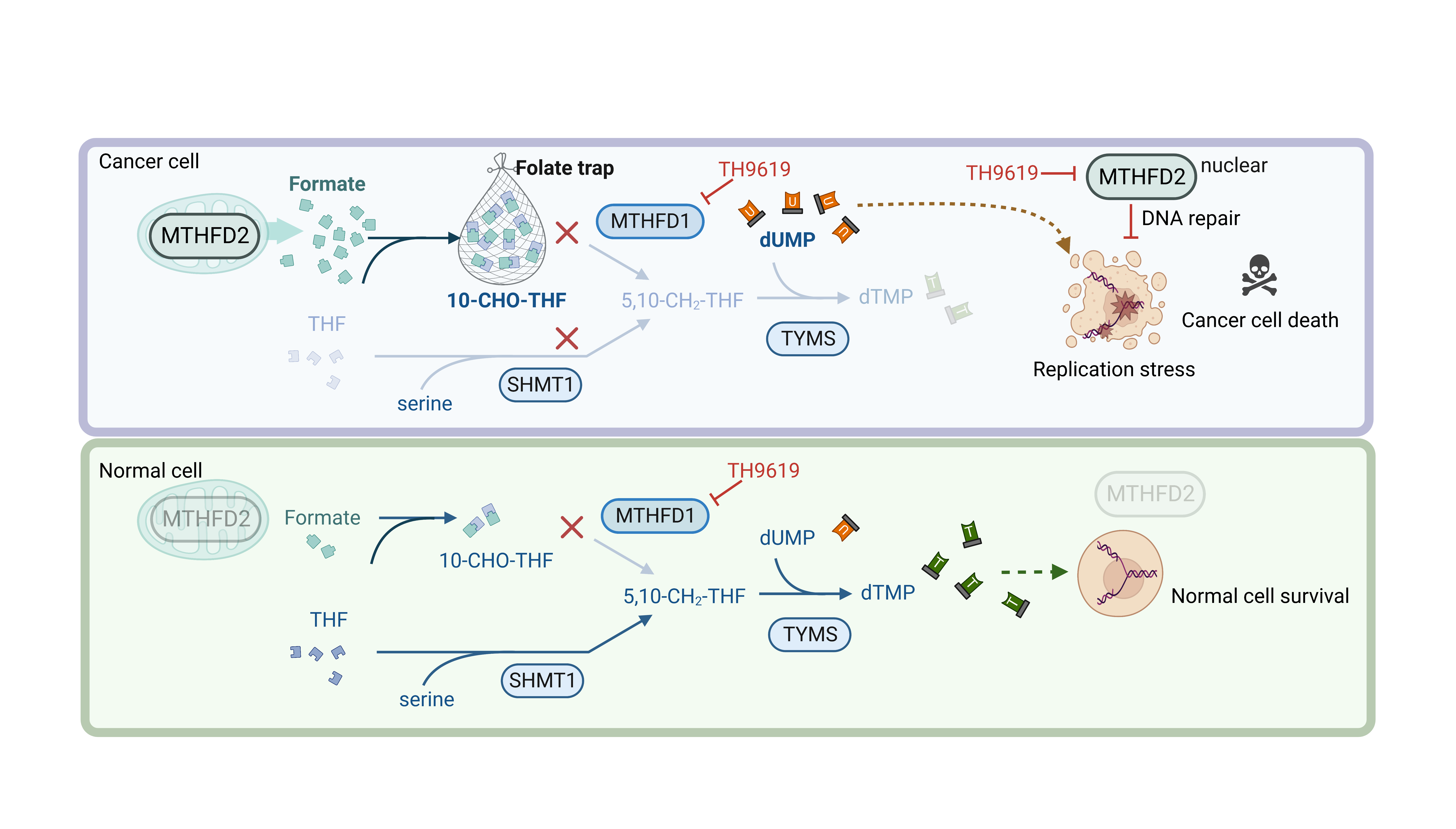
One-carbon Therapeutics to Present Trial-in-Progress Poster on ODIN Phase 1/2 Study ofTH9619 at the 2026 ASCO Annual Meeting
Solna, Sweden, May 29th, 2026. One-carbon Therapeutics AB, a clinical-stage biotechnology company pioneering first-in-class cancer therapies that exploit cancer’s oncogene addition, today announced that a